Are my Eyedrops Safe?


April 13, 2023
Eye doctors and patients across the globe are on high alert with a very important question that’s seemingly never broken ground before: “Are my eyedrops safe?” This post will cover drop composition, use, safety, and our best tips.
The PSA
In recent weeks, the press has highlighted several devastating cases of eye drop related infections resulting in vision loss, enucleation (removal of an eye), and even deaths. Our hearts go out to the patients and families affected, and our practice is fully committed to heightened surveillance, detailed communication with our patients and vendors, and science backed education on this topic.
*UPDATED list as of 11/16/23: Read more from the FDA here.
The following products have been recalled:
- EzriCare and Delsam Pharma Artificial Tears Lubricant Eye Drops.
- Delsam Pharma Artificial Eye Ointment.
- Apotex Brimonidine Tartrate Ophthalmic Solution, 0.15%.
- Pharmedica “Purely Soothing,” 15% MSM Drops.
- CVS Health Brand Lubricant/Lubricating and Muti-Action Relief Eye Drops
- Leader Brand (Cardinal Health) Irritation Relief, Lubricant and Dry Eye Relief Eye Drops
- Rugby Brand (Harvard Drug Group) Lubricating Eye Drops
- Rite Aid Brand Relief, Gel and Lubricating/Lubricant Eye Drops
- Target Up & Up Brand Lubricating and Dry Eye Relief Eye Drops
- Velocity Pharma Brand Lubricant Eye Drops
- Walmart Equate Lubricant Eye Drops
CVS, Target and Rite Aid are removing the products from their stores. At least two of these products are found as multi-dose preservative free bottles and all four have been associated with a rare strain of extensively drug resistant pseudomonas aeruginosa. While Pseudomonas is a common culprit of eye infections, this particular strain is novel and adept at penetrating into the cornea, and it does not respond well to first-line antibiotics.
Are my eyedrops safe?
In general, yes, but there are some variables that influence the safety and sterility of drops. A 2022 review paper looked at more than 100 articles over a 30-year period and concluded the following factors to be most commonly associated with risk of microbial contamination in eye drops: preserved versus preservative-free formulations, extended duration of use, improper administration technique, bottle squeezability, and age-related physical difficulties.1 Interestingly a separate study looked at instillation angle and found an angle of 45 degrees instead of 90 degrees is also significantly correlated with greater contamination rates.
Eye drops made with preservatives are formulated specifically to help ward off pesky pathogens like pseudomonas, and single-dose preservative free vials have a narrow tip specialty designed to keep these bugs out during a short duration of use. A number of studies have concluded however preservative-free eye drops in bottled form are at greater risk of contamination by potentially pathogenic micro-organisms so these may be wise to avoid for now. More on preservatives next.
While pharmaceutical companies strive to ensure the sterility of the eye drop tip, cap and contents and most products undergo extensive testing, since the recall, a number of manufacturers have pulled their drops from the shelves as an added safety measure. Our practice is happy to help answer any questions about specific drops you may be using.
So what’s an eye drop made out of anyway?
Eyedrops contain a combination of a demulcent for lubrication, an emollient for tear stabilization, one or more preservatives to increase shelf-life and combat bacteria, and buffers & electrolytes to improve comfort.(3)
- Demulcents: Water-soluble polymer that helps to protect and lubricate the mucous membranes of the eye. Examples: Carboxymethylcellulose, Hydroxymethylcellulose, Propylene Glycol
- Emollients: Lipid based emulsions are used to increase the lipid layer thickness, help stabilize the tear film, and reduce evaporation. Examples: Mineral oil, Castor oil, and Flaxseed oil.
- Preservatives: Designed to decrease the growth of bacteria once the bottle has been opened. Examples: Benzalkonium chloride (BAK), polixetonium, polyquaternium (Polyquad), OcuPure, sodium chlorite
- Other Ingredients: Added for comfort and to avoid stinging upon installation. Examples: Buffers and electrolytes.
What do Artificial Tears do?
Artificial tears support the tear film. Eye drops are designed to supplement the natural coating of tears that lives on the surface of the eyes. We call this the ‘tear film’. It’s the first interface that light passes through before making it to the cornea, lens and beyond. A healthy tear film has three intermingled components of mucin, aqueous and lipid that live in delicate harmony (Figure 1). Tear film stability is crucial in ensuring one’s best vision and is one of the most important assessments our doctors perform.
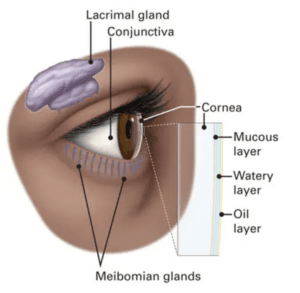
Figure 1. Components of the ocular surface ecosystem. The tear film is composed of a mucous layer, aqueous layer (watery), and a lipid (oil) layer.(4)
How often, and how much is too much?
Artificial tears can safely be used 4 to 6 times per day, with more frequent use often recommended perioperatively. More is not always better: overuse of artificial tears can wash out the eye’s natural tears and influence the stability of the tear film. *A good rule of thumb for lubricating artificial tears is 4-6x per day, unless otherwise directed.
So, are preservatives good or bad?
While preservatives help artificial tears last longer in the bottle and fight bacteria and other pathogens, some patients find the additives can irritate their eyes, leading to a hypersensitivity reaction called “medicamentosa”. Like many things, preservatives are okay in moderation. Talk to your doctor about the best type of drops and most effective dosing for your eyes.
What if drops aren’t cutting it?
Alternatives to drops are numbered and can include a combination of the following:
- Omega 3’s
- Eye Masks or Moisture Goggles
- Oral or Topical Medications
- Lubricating Ointment
- Punctal Plugs – temporary or permanent
- Autologous Serum
- Amniotic Membranes
- Neurostimulation Devices
- Eyelid and Meibomian Gland Treatments (microblepharoexfoliation, thermal pulsation, or intense pulse light)
Tips to take home
*If you’re using an artificial tear and you’re noticing something does not feel right, contact your eye doctor right away.
*Symptoms of extreme pain, light sensitivity, blurry vision and signs of redness or yellow, green or clear discharge are cause for concern, and should be immediately addressed.
*Always check expiration dates on unopened bottles. For opened bottles, use a Sharpie to write the date of opening on the bottom of the bottle – most pharmaceutical companies recommend unused drops be discarded after 28-30 days.
*Place the cap back on artificial tears and other bottled drops including contact lens solution to prevent cross contamination
*When possible, avoid keeping eye drops in excessively warm or moist environments.
*Patients who are immunocompromised and patients who wear contact lenses should take extra precautions.
To stay up to date on the CDC’s most current updates visit: Outbreak of Extensively Drug-resistant Pseudomonas aeruginosa Associated with Artificial Tears | HAI | CDC.
References:
1. Iskandar K, Marchin L, Kodjikian L, Rocher M, Roques C. Highlighting the Microbial Contamination of the Dropper Tip and Cap of In-Use Eye Drops, the Associated Contributory Factors, and the Risk of Infection: A Past-30-Years Literature Review. Pharmaceutics. 2022; 14(10):2176. https://doi.org/10.3390/pharmaceutics14102176
2. da Costa AX, Yu MCZ, de Freitas D, et al. Microbial Cross-contamination in Multidose Eyedrops: The Impact of Instillation Angle and Bottle Geometry. Transl Vis Sci Technol. 2020;9(7):7. Published 2020 Jun 5, doi:10.1167/tvst.9.7.7
3. https://www.reviewofoptometry.com/article/artificial-tears-what-matters-and-why
4. https://www.aao.org/eye-health/anatomy/tear-film-3

Dr. Matthew R. Sharpe